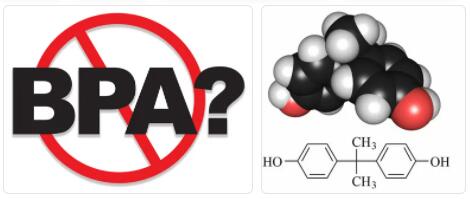
Short for Bisphenol A according to abbreviationfinder, BPA is a chemical product that has been used for many years as a component for the manufacture of polycarbonate and epoxy-phenolic resins. This substance is currently authorized for the manufacture of plastic materials by Regulation (EU) 10/2011 of the Commission, of January 14, 2011, on plastic materials and objects intended to come into contact with food.
Polycarbonate is a type of transparent rigid plastic that is used both to make food containers and many other objects not related to food with which we are in contact every day, such as cash register receipts made of thermal paper, CDs or DVDs, toys, cosmetics, etc.
For their part, epoxy-phenolic resins are used in coatings and coatings for canned foods and food and beverage containers.
Synthesis
Bisphenol-A was first synthesized by the Russian chemist Aleksandr Dianin in 1891. It is prepared by the condensation of acetone (hence the suffix ‘-A’)5 with two equivalents of phenol. The reaction is catalyzed by an acid, such as hydrochloric acid (HCl) or a sulfonated polystyrene resin. A large amount of phenol is normally used to ensure its complete condensation. A large number of ketones undergo analogous condensation reactions. This method is efficient and the only by-product is water.
Applications
Bisphenol-A is used primarily to make plastics. Products containing bisphenol-A have been on the market for more than fifty years. It is a key monomer in the production of epoxy resin and in the most common form of polycarbonate plastic. The plastic polycarbonate, which is transparent and nearly shatterproof, is used to make a wide variety of common products, including baby bottles and water bottles, sports equipment, medical and dental devices, dental composites and sealants, organic eyeglasses, CDs and DVDs, and various appliances.
It is also used in the synthesis of polysulfones, polyether ketones, as an antioxidant in some plasticizers, and as a polymerization inhibitor in PVC. Epoxy resins containing Bisphenol-A are used as a coating on almost all food and beverage cans,12 however, due to health concerns, in Japan the epoxy coating was replaced by a polyester film. BPA is also a precursor to a flame retardant, tetrabromobisphenol A, and is used as a fungicide. In addition, BPA is a color developer for thermal paper15 and for NCR paper. Products passed in BPA are used in foundry molds and as coatings for water pipes.
Identification in plastics
There are 7 types of plastic used in packaging. Type 7 contains all the “others”, and some of these, like polycarbonate (sometimes identified by the letters ‘PC’ next to the recycling symbol) and epoxy resins, are made from the Bisphenol-A monomer.
Type 1, 2, 4 and 5 plastics
Type 1 (PET), 2 (HDPE), 4 (LDPE) and 5 (Polypropylene) plastics do not use Bisphenol-A during their polymerization or packaging formation.
Type 3 plastics
Type 3 plastics (PVC) may also contain bisphenol-A as an antioxidant in plasticizers.
Type 7 plastics
ABS (Acrylonitrile-butadienestyrene) is a hard plastic used in pipes, car fenders and toys. Its production uses butadiene and styrene in addition to acrylonitrile, a highly toxic product. Given its very complex chemical composition, ABS is extremely difficult to recycle. Compact discs and 19 liter water jugs are usually made of polycarbonate (PC). PC does not require additives, but its production uses phosgene and solvents such as chloroethane and chlorobenzene as well as bisphenol-A, a frequently used endocrine disruptor. Processes for producing PC without these compounds are currently being explored. Some PC recovery processes have been designed to produce lower quality products.
Type 6 plastics
Polystyrene (PS). Plastic foam (better known by the brand name “Styrofoam”). Includes hot drink cups, fast food clamshells, egg cartons and meat trays. PS represents approximately 11% of all plastics. All other plastic resins or mixtures of those indicated above in the same product. These plastics represent approximately 4 % of all plastics.
affected
The main affected are mostly babies, both in their embryonic stage and in lactation, because that is when hormones play a fundamental role. It is in these moments of his life, when his mother transfers to him, either through the umbilical cord or through milk, all these hormones that have been accumulating in her body.
Toxicity
Bisphenol A is an endocrine disruptor. It is capable of causing imbalances in the hormonal system at very low concentrations with possible repercussions on health. Its toxic effects are due to the consumption of food that has been contaminated by contact with materials that contain this substance, such as containers, cans or containers of a very diverse kind. The wide distribution of products with bisphenol A, especially in developed countries, causes continuous exposure of the population, affecting all ages (from fetuses to the elderly). The continuous presence of this disruptor in the body has been related to an increased risk of various organic disorders.
toxic effects
Numerous alterations have been associated with bisphenol A in living beings, based on a deregulation of the endocrine system and the corresponding hormone production:
Effects on the male reproductive system
Numerous works refer to an alteration of spermatogenesis that conditions a decrease in sperm levels, testosterone and, in general, male fertility. In addition, other studies suggest a change in sexual behavior.
Effects on the female reproductive system
In women, changes occur in the maturation of the oocytes, decreasing their number and quality. There is also some study that relates exposure to bisphenol A with negative effects on the endometrium, appearance of polycystic ovaries, abortions and premature births. On the other hand, in animals there is evidence of ovarian cysts, endometriosis, early puberty and involvement of the hypothalamic-pituitary-gonadal axis. Bisphenol A can modify the activity of maternal steroids in the embryos of vertebrate animals.
Effects on the brain and behavior
Various animal trials have confirmed the effect of bisphenol A on neuron differentiation, alteration of glutamine- and dopamine-mediated systems, and changes in estrogen receptor expression. It has also been linked to possible changes in maternal behavior (less attention to offspring), anxiety, reduced exploratory behavior and a feminization of males. In humans, changes have been established that include hyperactivity, increased aggressiveness, increased susceptibility to addictive substances, and problems in both learning and memory.
Effects on metabolism and cardiovascular system
Associations of an increased concentration of bisphenol A with cases of various heart diseases and hypertension have been established. In addition, exposure to bisphenol A leads to increased blood lipids, weight gain, and increased lipogenesis. It can also influence the appearance of type II diabetes mellitus by increasing insulin resistance and the number of fat cells.
Effects on the thyroid
Animal studies conclude that bisphenol A is capable of affecting thyroid function, behaving as an antagonist of thyroid hormone. In amphibians, this effect translates into an inhibition of their metamorphosis. Thyroid involvement also affects rodents. In the case of humans, not enough studies have been carried out to allow conclusive results to be drawn.
Effects on the immune system
An induction of T lymphocytes and an increase in the production of cytokines have been shown in experimental animals, thus favoring allergic processes.
Effects on the gut
Possible inflammation and alteration of intestinal permeability in animals.
Carcinogenic effects
When bisphenol A is metabolized by hydroxylation and subsequent oxidation, it forms an orthoquinone that can establish covalent bonds with DNA and develop mutagenic and teratogenic effects. The mutagenic effects could be the initiators of several carcinogenic processes associated with bisphenol A:
- Prostate cancer
The estrogenic activity of the substance can lead to an increase in prostate size and a decrease in the size of the epididymis.
- Breast cancer
Increased mutagenic and carcinogenic susceptibility has been detected at the level of mammary cells in women due to estrogenic stimulation of the development and division of the mammary glands.